Abstract
Introduction: Advances in AML research have expanded the treatment landscape to newer approved therapies targeting disease-related mutations. One key molecular marker that has been identified to be prognostic in AML is the FMS-like tyrosine kinase 3 (FLT3) mutation (FLT3+). While the treatment landscape for patients (pts) with FLT3+ AML expands, there is limited real-world evidence around the impact of FLT3+ targeted therapies, especially in the R/R setting. The FLT3+ R/R AML pt population is uncommon and shown to have a poor prognosis. This study examines baseline characteristics, treatment patterns, overall survival (OS), and time to next treatment (TTNT) among FLT3+ R/R AML pts using a heavily community focused real-world dataset.
Methods: This ongoing retrospective longitudinal study included pts, age ≥ 18 years, with a confirmed diagnosis of AML, FLT3+ status, and at least one R/R event between January 2015 and January 2022. All pts received at least first line (1L) of treatment (tx) following 1st R/R event after AML diagnosis (1L tx in R/R AML). Pts were identified from the ConcertAI Oncology Dataset, which draws electronic medical records from a wide range of principally community oncology practices throughout the U.S. Pts were grouped into mutually exclusive groups based on the systemic treatments they received after first R/R diagnosis: a) FLT3 tyrosine kinase inhibitor with or without chemotherapy combination (FLT3 TKI), b) High intensity chemotherapy (HIC), c) Low intensity chemotherapy (LIC), or d) Other. Within the FLT3 TKI group, the subgroup of pts receiving gilteritinib in 1L tx as monotherapy or in combination was further examined. Kaplan-Meier methods were used to determine OS and TTNT. OS was measured from the start of 1L tx, and pts without evidence of death were censored at the last observed visit. TTNT was measured from start of 1L tx to start of 2L tx, and pts with no 2L tx were censored at the last observed visit.
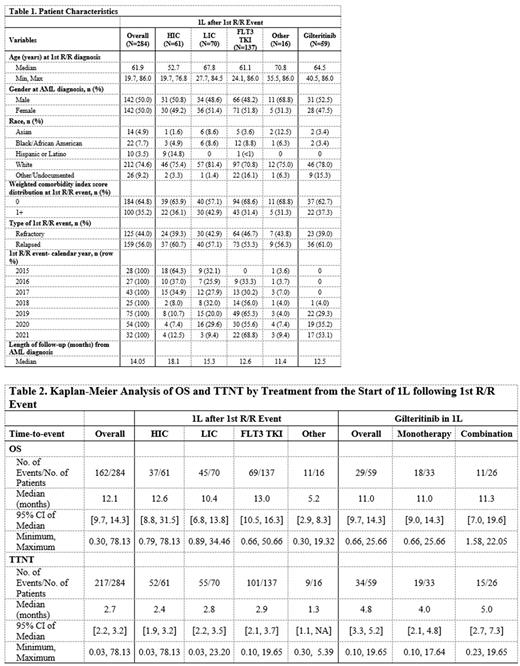
Results: This study included 284 pts (50.0% male) who initiated 1L tx in R/R AML. About 21.5% (n=61) received HIC, 24.6% (n=70) received LIC, 48.2% (n=137) received a FLT3 TKI, and 5.6% (n=16) received Other tx. Of the subset of pts receiving a FLT3 TKI in 1L tx, 43.1% (n=59) received gilteritinib. Overall, most pts were White (74.6%, n=212) with a median age of 61.9 at 1st R/R event. Median age was 52.7 years for HIC group, 67.8 years for LIC group, 61.1 years for FLT3 TKI group, and 70.8 years for pts in Other group. Among pts receiving gilteritinib in 1L tx, median age was 64.5 years. Median follow-up time was 18.1 months for HIC pts, 15.3 months for LIC pts, 12.6 months for FLT3 TKI pts, and 11.4 months for pts receiving Other tx. Additional pt characteristics by treatment are shown in Table 1. Use of gilteritinib in 1L tx per R/R diagnosis year increased from 4.0% in 2018 to 53.1% in 2021, while use of HIC in 1L tx decreased from 64.3% in 2015 to 12.5% in 2021. More than half (55.9%) of pts receiving gilteritinib had it as monotherapy. Median OS (95% CI) across pts with 1L tx in R/R AML was 12.6 (8.8, 31.5) months among HIC, 10.4 (6.8, 13.8) months among LIC, 13.0 (10.5, 16.3) months among FLT3 TKI, and 5.2 (2.9, 8.3) months among Other tx groups. Pts receiving gilteritinib in 1L tx had a median OS of 11.0 (9.7, 14.3) months; 11.0 (9.0, 14.3) months among gilteritinib monotherapy and 11.3 (7.0, 19.6) among gilteritinib combination therapy. Median TTNT (95% CI) was 2.4 (1.9, 3.2) months among HIC, 2.8 (2.2, 3.5) months among LIC, and 4.8 (3.3, 5.2) months among gilteritinib treated pts. Further OS and TTNT results are shown in Table 2.
Conclusion: This study provides insight into real-world practices for the treatment of FLT3+ R/R AML. In community oncology settings, increased use of gilteritinib was seen in recent years (2019-2021) while use of HIC has been decreasing. More than half of the gilteritinib pts in this study received monotherapy. Median OS for gilteritinib monotherapy pts from this study (11.0 months) was comparable to results from the ADMIRAL trial, where median OS for gilteritinib treated pts was 9.3 months. Given the evolving treatment landscape for FLT3+ AML pts, this study demonstrates the need for continued observation of treatment patterns and clinical outcomes among real-world pts.
Disclosures
Zeidan:Novartis, Cardiff Oncology, Pfizer: Other: Travel Support; Pfizer, Boehringer-Ingelheim, Trovagene, Incyte, Takeda, Amgen, Aprea, Gilead, Kura, Loxo Oncology, Otsuka, Jazz, Agios, Acceleron, Astellas, Daiichi-Sankyo, Cardinal Health, Taiho, Seattle Genetics, BeyondSpring, Ionis, Epizyme, Janssen, Syndax, Genentec: Consultancy, Honoraria, Other: Advisory Boards; Celgene/BMS, Novartis, AbbVie, Gilead, Kura, Loxo Oncology, Geron: Other: Clinical Trial Committee; Gilead, Kura, Loxo Oncology: Consultancy, Honoraria, Other: Clinical Trial Committee; Celgene/BMS, Novartis, Cardiff Oncology, AbbVie, Pfizer, Boehringer-Ingelheim, Trovagene, Incyte, Takeda, Amgen, Aprea, Astex, Pfizer, Medimmune/AstraZeneca, ADC Therapeutics: Research Funding; Celgene/BMS, Novartis, Cardiff Oncology, AbbVie: Consultancy, Honoraria, Other: Advisory Board; Jazz, Agios, Acceleron, Astellas, Daiichi Sankyo, Cardinal Health, Taiho, Seattle Genetics, Beyondspring, Gilead, Kura, Tyme, Janssen, Syndax, Geron, Ionis, Epizyme: Consultancy, Honoraria; Astex, Medimmune, Astrazeneca, ADC Therapeutics: Research Funding; Celgene/BMS, AbbVie, Pfizer, Boeringer-Ingelheim, Trovagene, Cardiff Oncology, Incyte, Takeda, Novartis, Aprea, Amgen, Otsuka: Consultancy, Honoraria, Research Funding. Gautam:Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA: Research Funding; Astellas: Research Funding; ConcertAI: Current Employment, Other: Medical publication support. Yu:Astellas: Research Funding; ConcertAI: Current Employment, Other: Medical publication support; Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA: Research Funding. Lan:Astellas: Research Funding; ConcertAI: Current Employment, Other: Medical publication support; Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA: Research Funding. Grinblatt:AbbVie: Consultancy; Astellas Pharma: Consultancy, Research Funding; AstraZeneca: Consultancy; Bristol Myers Squibb: Consultancy; ConcertAI: Other: Medical publication support. ElSouda:Astellas: Current Employment, Research Funding; ConcertAI: Other: Medical publication support. Spalding:Astellas: Current Employment, Research Funding; ConcertAI: Other: Medical publication support. Block:Astellas: Current Employment, Research Funding; ConcertAI: Other: Medical publication support. Touya:ConcertAI: Other: Medical publication support; Astellas: Current Employment, Research Funding. Pandya:ConcertAI: Other: Medical publication support; Astellas: Current Employment, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal